 When the electrons move or shift, they create electrically positive and negative areas. Or electrons can shift around inside a molecule. Other times, they might jointly share the electrons with that neighbor. Atoms can give up electrons to a neighboring atom. 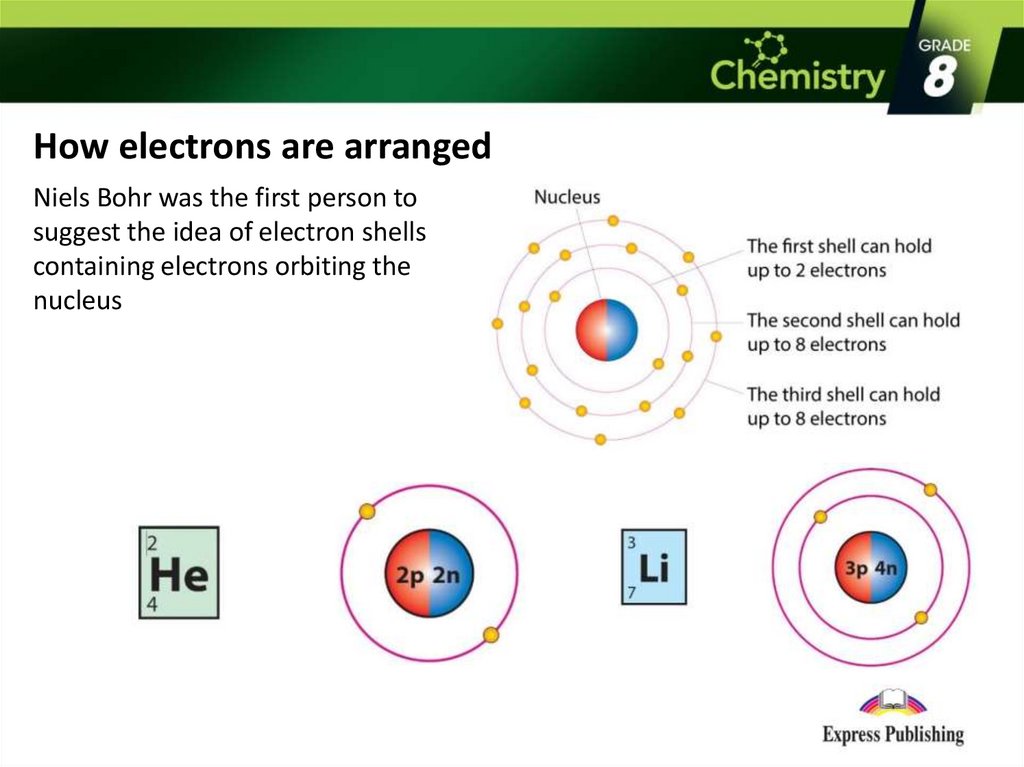
How they behave will control the properties of a bond. (Positively charged protons and electrically neutral neutrons are the others.) Electrons carry a negative charge. But electrons control all bonds, no matter what type.Įlectrons are of one the three primary sub-atomic particles that make up atoms. Intra- and inter-bonding are further divided into different types. (Intra means within.) Those that attract one compound to another are known as inter bonds. Those that hold one building block to another inside a compound are known as intra bonds. Can we use a material as a lubricant? Once again, check out its bonds.Ĭhemical bonds broadly fall into two categories. Those bonds also will determine if a substance conducts electricity. To know if a material dissolves in water, for instance, we look to its bonds. They also determine the structure - and therefore the properties - of all substances. 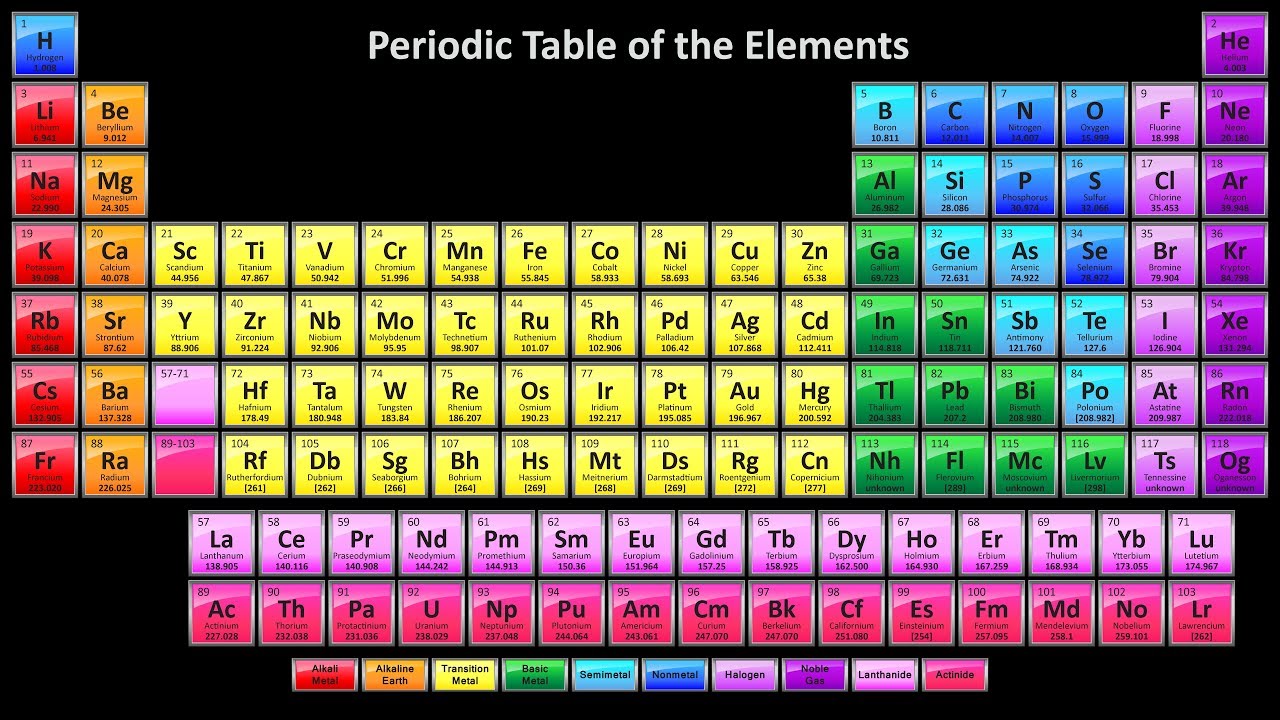
Quite simply, they hold our universe together. 
Additional, weaker types of bonds can attract one compound to another. Within the compound, bonds are what “glue” each of the blocks together. With enough jars, you can use the blocks to build anything - as long as you follow a few simple rules. And each represents an atom of a different element on the periodic table. Every type is a slightly different color, size and shape. Imagine a glass jar holding 118 types of building blocks. 
0 Comments
Leave a Reply.AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
